
Ultimate Guide to Biodegradable Smoking Filters
Biodegradable smoking filters use coconut-shell charcoal, unbleached paper and ceramic tips to reduce toxins and decompose in 1–3 years.

*Image is generated for the purpose of this article and does not present a Charco product.
Activated charcoal has been used in medicine for thousands of years, starting with ancient Egyptians who utilized it for wound care and water purification. Its unique porous structure, created through high-temperature activation, allows it to trap toxins effectively. Today, it’s a trusted treatment for poisonings and overdoses, reducing toxin absorption by up to 88.4% when administered quickly. Historical milestones include its use by Hippocrates in 400 B.C., advancements in adsorption science during the 18th and 19th centuries, and its role in modern drug purification and emergency medicine. From ancient remedies to pharmaceutical-grade materials, activated charcoal remains a key tool in healthcare.
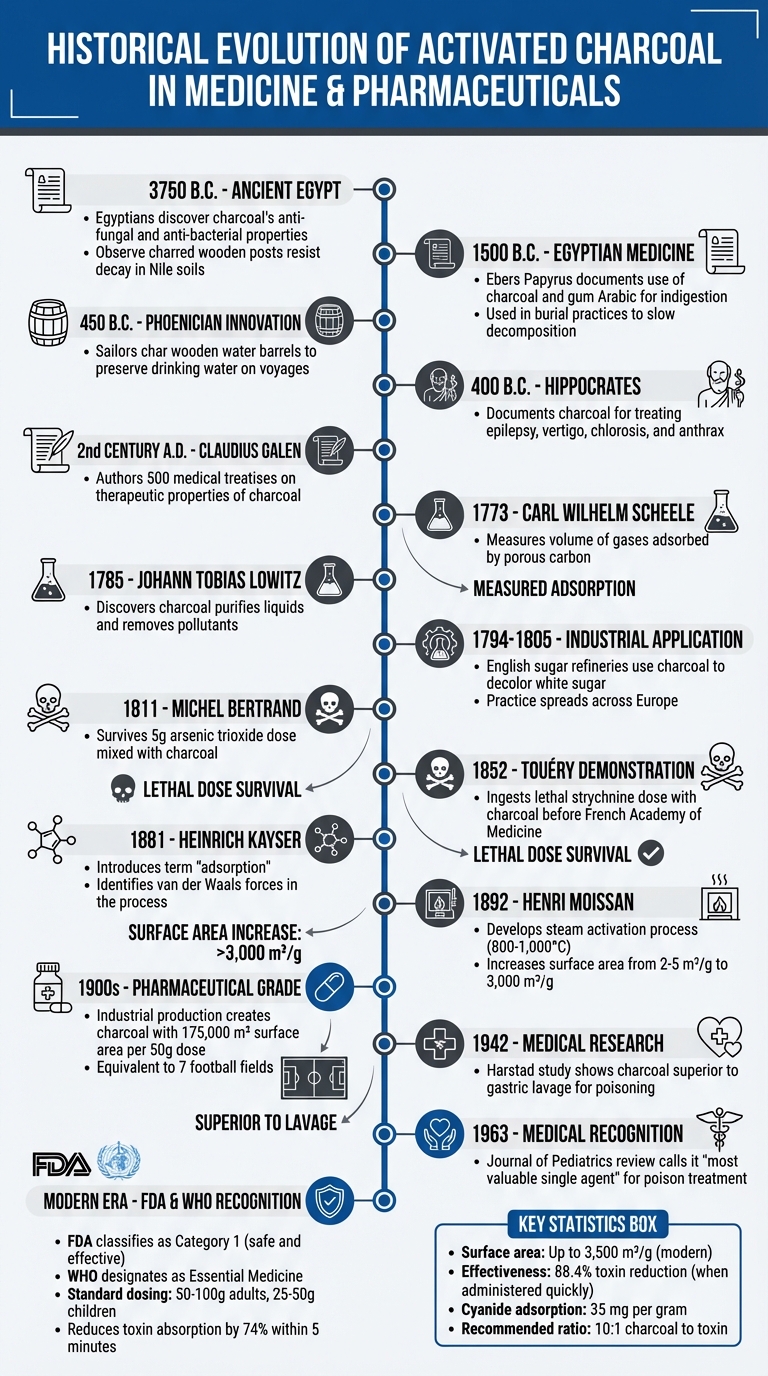
Timeline of Activated Charcoal in Medicine: From 3750 BC to Modern Pharmaceuticals
The Egyptians were among the first to recognize the practical benefits of charcoal, dating back to 3750 B.C.. They observed that wooden posts charred by fire resisted decay in the soils of the Nile, hinting at charcoal’s anti-fungal and anti-bacterial qualities. This discovery marked the beginning of its various applications.
By 1500 B.C., the Ebers Papyrus detailed how Egyptians used "Egyptian ink" – a mix of charcoal and gum Arabic – to treat indigestion. Charcoal also played a role in their burial practices, where they encased bodies in a combination of charcoal and sand to slow decomposition.
Charcoal’s ability to purify was further demonstrated by ancient sailors around 450 B.C.. The Phoenicians charred the interiors of wooden water barrels to preserve drinking water during long voyages, ensuring its freshness for extended periods.
These early uses not only showcased charcoal’s versatility but also influenced medical and practical applications in later civilizations.
Building on Egyptian knowledge, the Greeks expanded charcoal’s role in medicine. Around 400 B.C., Hippocrates documented its use for treating conditions like epilepsy, vertigo, chlorosis (a severe iron-deficiency anemia), and even anthrax. This marked a shift toward using charcoal for internal and neurological ailments, showcasing its growing importance in ancient medicine.
In the 2nd century A.D., Roman physician Claudius Galen furthered these practices. He authored nearly 500 medical treatises, many of which discussed the therapeutic properties of charcoals derived from both plants and animals. While the concept of "adsorption" wasn’t formally defined until 1881, these early practitioners understood charcoal’s effectiveness through careful experimentation and observation.
These ancient insights laid a foundation for the broader medical applications of activated charcoal in later eras.
In 1773, Swedish chemist Carl Wilhelm Scheele took a closer look at charcoal’s ability to attract and hold gases. He measured the volume of gases adsorbed by porous carbon, providing a clearer picture of its properties. A few years later, in 1785, Russian chemist Johann Tobias Lowitz discovered that charcoal could also purify liquids. By removing unpleasant tastes, odors, and organic pollutants, it even proved effective in decolorizing solutions. This discovery quickly found practical use, and by 1794, English sugar refineries were using charcoal to decolor white sugar – a practice that spread across Europe by 1805.
The success of charcoal in sugar refining spurred further research into its properties. In 1865, John Hunter delved deeper into the adsorptive qualities of activated carbon. Then, in 1881, German physicist Heinrich Kayser introduced the term "adsorption" to describe how gases adhere to the surface of a solid. He also identified the role of van der Waals forces in this process.
These early breakthroughs laid the groundwork for significant advancements in processing techniques that would drastically improve the performance of activated charcoal.
The transition from understanding charcoal’s chemical properties to refining its production methods marked a major milestone. In 1892, French chemist Henri Moissan developed the steam activation process, which revolutionized the industry. By heating charcoal in the presence of steam at temperatures between 800°C and 1,000°C (1,472°F to 1,832°F), this method opened up previously inaccessible pores, dramatically increasing their number and size. To put it in perspective, standard charcoal offers a surface area of 2–5 m²/g, while steam-activated charcoal boasts an impressive 3,000 m²/g. This enhanced porosity made it far more effective at capturing gases and toxins, cementing its role in pharmaceutical applications.
Moissan’s steam activation process became the gold standard and remains widely used today. His broader contributions to science earned him the Nobel Prize in Chemistry in 1906.
These innovations not only improved the adsorptive power of activated charcoal but also established it as a key material in modern drug production and other industries.
In the early 1900s, industrial advancements brought significant improvements to the production of pharmaceutical-grade charcoal. By exposing charcoal to extreme heat – ranging from 600°F (315°C) to 1,652°F (900°C) – its adsorptive capacity was dramatically increased. To put this into perspective, a 50-gram dose of this enhanced charcoal could offer an adsorptive surface area of about 175,000 square meters. That’s roughly equivalent to the size of seven football fields.
The groundwork for these advancements can be traced back to earlier experiments. In 1811, French pharmacist Michel Bertrand ingested 5 grams of arsenic trioxide – a dose that would typically be fatal – mixed with charcoal and survived. Similarly, in 1852, chemist Touéry consumed a lethal dose of strychnine combined with charcoal in front of the French Academy of Medicine, emerging unharmed. These dramatic demonstrations highlighted charcoal’s potential as an antidote. Over time, further refinements led to the creation of "superactivated" charcoal, which boasted surface areas of 2,800–3,500 square meters per gram. Research confirmed its ability to adsorb up to 35 milligrams of cyanide per gram. As a result, a standard pharmaceutical guideline emerged, recommending a ratio of 10 parts activated charcoal to 1 part toxin by weight. Some studies, however, suggested that a 40:1 ratio might be more effective for larger amounts of ingested toxins.
These findings solidified activated charcoal’s role as a reliable poison antidote, ultimately setting the stage for its use in critical medical applications during wartime.
During the World Wars, activated charcoal became an essential component of gas mask filters, effectively neutralizing chemical agents. While early medical practices often relied on techniques like gastric lavage and ipecac to treat poisoning, research began to reveal the superior efficacy of charcoal. For instance, a 1942 study conducted by Harstad and Danish colleagues demonstrated that gastric lavage retrieved only trace amounts of phenobarbital, highlighting the greater effectiveness of activated charcoal in such cases.
The medical community’s appreciation for activated charcoal grew substantially after a 1963 review published in the Journal of Pediatrics by L. E. Holt Jr. and P. H. Holz. They described its wide-ranging capabilities, stating:
"This agent, presently somewhat neglected, has a wide spectrum of activity and when properly used is probably the most valuable single agent we possess".
This recognition underscored activated charcoal’s versatility and cemented its status as a cornerstone in poison treatment and medical research.
Activated charcoal remains a go-to treatment for acute poisonings and overdoses, thanks to its ability to adsorb toxins through its porous structure, preventing them from entering the bloodstream. Standard formulations offer a surface area ranging from 800 to 1,200 m²/g, while advanced versions can reach up to an impressive 3,500 m²/g.
Timing plays a huge role in its effectiveness. If administered within 5 minutes of toxin exposure, activated charcoal can reduce absorption by about 74%. However, its efficacy drops significantly over time – 47.3% at 30 minutes, 40.1% at 60 minutes, and just 16.5% after 2 hours. Current dosing guidelines, refined through years of research, suggest 50–100 g for adults, 25–50 g for children aged 2–12, and 10–25 g for infants, typically following a 10:1 ratio of charcoal to toxin by weight. In severe cases, multiple doses can disrupt enterohepatic recirculation, speeding up the elimination of drugs like carbamazepine, dapsone, phenobarbital, quinine, and theophylline.
Beyond its role in emergency medicine, activated charcoal’s highly refined properties make it indispensable in pharmaceutical production.
In drug manufacturing, activated charcoal is used to remove impurities, ensuring medications meet stringent quality and purity standards. Its selective adsorption capabilities allow it to trap large-molecule impurities while leaving the desired compounds intact. This process is critical for meeting the purity requirements set by regulatory agencies like the FDA, which classifies activated charcoal as Category 1 ("safe and effective") for treating acute toxic poisoning.
The transformation of activation techniques over time highlights the evolution of activated charcoal’s effectiveness. Here’s a breakdown of how methods have advanced:
| Feature | Historical Methods (Pre-1900s) | Modern Steam Activation | Modern Chemical Activation |
|---|---|---|---|
| Process | Basic charring | Oxidation at 1,472–1,832°F (800–1,000°C) using steam or CO₂ | Treatment with phosphoric acid or zinc chloride at lower temperatures |
| Raw Materials | Wood (oak, beech) or animal bones | Coconut shells, coal, or wood | Wood, peat, or sawdust |
| Surface Area | Limited to natural pores | Extremely high; creates a network of micro and macropores | High; tailored pore sizes for specific needs |
| Toxin Binding Capacity | Minimal; relies on physical filtration | Up to 35–1,800 mg per gram depending on the substance | Optimized for large-molecule impurities |
These advancements demonstrate how far activated charcoal has come, evolving from simple remedies into a cornerstone of modern pharmaceutical processes. Its journey from basic charring to cutting-edge activation methods reflects its ongoing importance in both medicine and manufacturing.
Activated charcoal has come a long way over the past 3,500 years, evolving from a rudimentary tool for wound care into an FDA-recognized treatment for poisoning. What started as basic charring techniques has grown into a sophisticated process that produces high-porosity charcoal, revolutionizing how we manage toxins and purify drugs.
This journey highlights our growing expertise in adsorption science. Between 1870 and 1920, steam activation methods transformed ordinary charcoal into a highly porous material, making it essential for applications like gastrointestinal decontamination, dialysis, and pharmaceutical production. These advancements set the stage for its expanded role in modern medicine.
Today, activated charcoal’s uses extend well beyond emergency settings. It plays a vital role in drug purification, wound care, hemoperfusion cartridges, and other specialized medical devices. Its designation as an "Essential Medicine" by the World Health Organization underscores its enduring significance in healthcare worldwide.
Its versatility has also spurred innovation across industries. From ancient water purification to cutting-edge drug refinement, activated charcoal remains a crucial tool. Products like Charco Filters, which incorporate coconut shell-based activated charcoal into eco-friendly filtration systems, illustrate how this ancient material continues to adapt to modern needs. These designs reflect the timeless value of activated charcoal’s ability to trap impurities while remaining biologically inert.
The history of activated charcoal demonstrates how scientific progress has transformed a simple natural remedy into a precise medical and industrial tool. Its ongoing evolution ensures that it will remain a cornerstone of emergency medicine and pharmaceutical manufacturing for years to come.
Activated charcoal works by adsorbing toxins onto its expansive surface area through physical forces. This action limits the absorption of harmful substances within the gastrointestinal tract, which is why it’s widely utilized in medical and pharmaceutical treatments.
Activated charcoal doesn’t work for everything. It’s ineffective against substances like alcohol, hydrocarbons (such as gasoline or kerosene), and corrosive chemicals like acids or alkalis. This is because these toxins have chemical properties that prevent activated charcoal from adsorbing them effectively.
Activated charcoal is most effective when taken quickly after exposure, as it works by rapidly binding to toxins or drugs in the gastrointestinal tract. To get the best results, it’s recommended to use it within 30 minutes of ingestion. This timing helps block absorption and supports the body in eliminating harmful substances.

Biodegradable smoking filters use coconut-shell charcoal, unbleached paper and ceramic tips to reduce toxins and decompose in 1–3 years.

From discarded shells to activated carbon: lower-emission production, waste-to-resource benefits, and uses in water and smoking filtration.

Cigarette filters can persist for years, fragment into microplastics, and only decompose fast in managed compost—material and disposal shape their harm.

Activated charcoal filters trap many gas-phase toxins, reduce throat irritation and preserve THC/CBD, but they don’t remove gases like CO or eliminate smoking risks.

Charcoal-driven smoke rituals blend spiritual meaning with practical purification—from Indigenous ceremonies to modern activated charcoal filters.

Coconut shell charcoal filters cut lifecycle emissions versus coal, but pyrolysis and pit charring remain the largest sources of carbon impact.